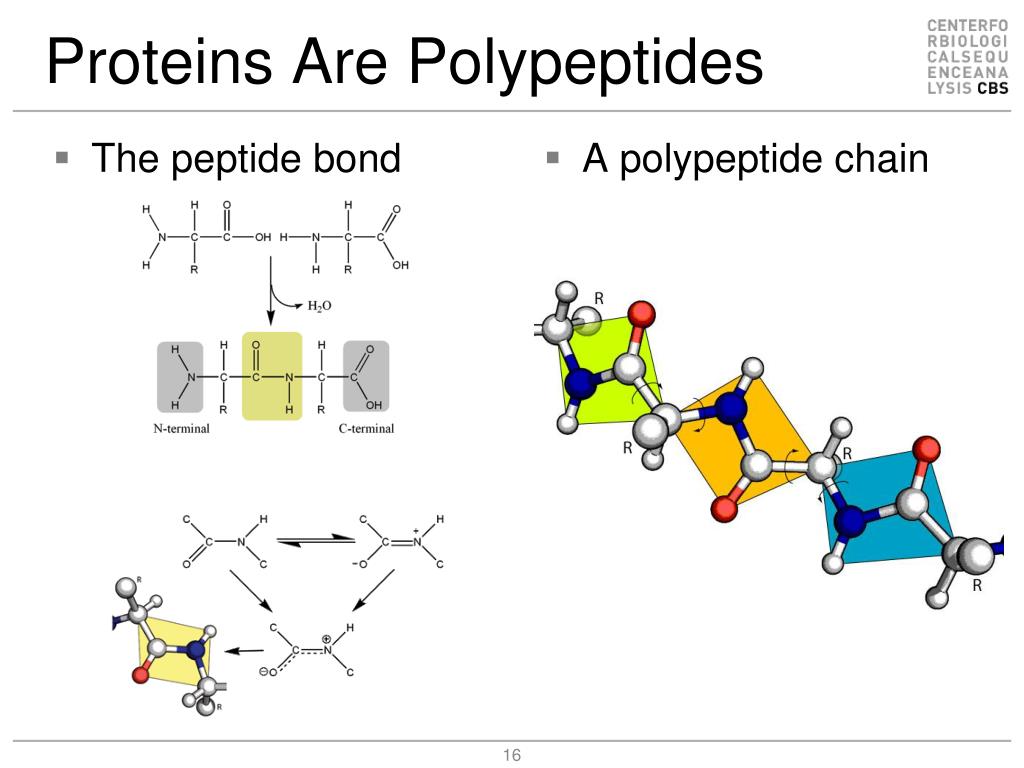
Random coils mean the irregular peptide chain. In the backbones of polypeptide chain, the structures which are different from the α-helix and β-sheet are called random coil. The polarization of the peptide chain is opposite for the neighboring chains. The N-end of all the peptide chains is in the same direction. The arrangement polarization of its peptide chain (N–C) is unidirectional. Along the long axis of peptide chain, there are repeated units. The hydrogen bonds are almost vertical to the long axis of peptide chains. In the β-sheets, all peptides join in the cross-linking between hydrogen bonds. These polypeptide structures are β-sheet. Hydrogen bond is formed by –NH and C=O on the neighboring peptide backbones. Two or more fully expended polypeptides cluster together laterally. Β-Sheet is another frequently occurrence structure. α-Helix tends to be stable because the hydrogen in NH and the oxygen in the fourth residue CO form hydrogen bond. Tightly curled polypeptide backbone forms the inner side of the stick the side chains expand outside in the form of helix. Local organization of protein backbone is α-helix, β-strand (which assembles into β-sheet), turn, and interconnecting loop. Various Kinds of Protein Secondary Structure Different classes of proteins have various functions. Conjugated proteins can also be subdivided into nucleoprotein, lipoprotein, glycoprotein and mucoprotein, phosphoprotein, hemoprotein, flavoprotein, and metalloprotein. Simple proteins can be subdivided into seven subclasses: albumin, globulin, glutelin, prolamine, histone, protamine, and scleroprotein. They can be divided into soluble fibrins and unsolvable fibrins. Comparatively, fibrins are less symmetric and look like thin sticks or fibers. Globins dissolve easily and can crystallize. Globins are more symmetric and similar to balls or ovals in shape. If proteins are completely composed of amino acids, these proteins are called simple proteins, such as insulin if there are other components, they are named conjugated proteins like hemoglobin.Īccording to the symmetry of proteins, proteins can be divided into globin and fibrin. 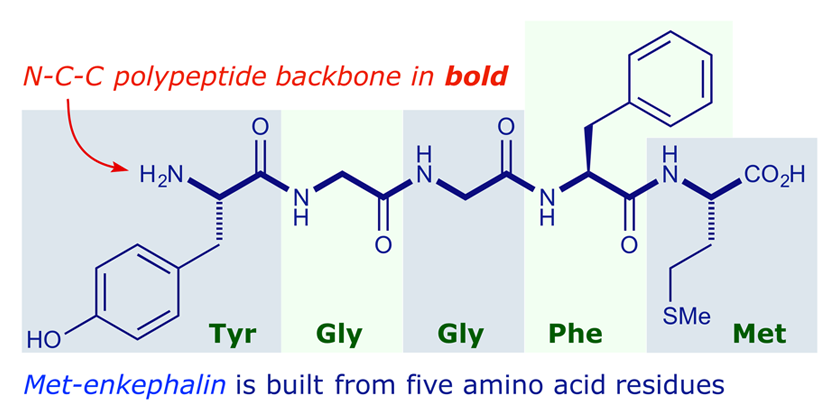
So, a likely scenario is that the proline is at the surface of the protein, providing a structural turn between two secondary structure elements (β strands or α helices), the serine is still close enough to the surface to interact with water, and the alanine is close enough to the interior of the protein to interact with other hydrophobic side chains.From the aspect of chemical structures of proteins, proteins can be classified into two classes. Proline residues have a restricted degree of conformational freedom because the side chain is covalently linked to the backbone nitrogen as well as the α-carbon. This peptide is made up of both polar and nonpolar amino acids, and one of the nonpolar amino acids is proline. The side chains are most likely buried in the interior of the protein, which would promote interactions with other hydrophobic side chains and avoid unfavorable interactions with the aqueous environment of the cytosol. This peptide is made up of nonpolar amino acids. This tripeptide is made up of entirely polar amino acids, which means it will most likely be found on the surface of the protein, interacting with the aqueous environment of the cytosol.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
